Welcome to the 125th edition of the Research Digest. This month’s collection reflects the continued strengthening of the evidence base across ME/CFS and related conditions, spanning biological mechanisms, lived experience, and illness trajectories. Together, the featured work reinforces the complex, multisystem nature of ME/CFS, the significant impacts on those most severely affected, and the growing momentum toward clearer biological understanding and improved recognition.
Contributing Digesters: Sarah, Anna, Shan & Simone.
We begin this year feeling very grateful for the additions to the Research Digest that have been shaped by the Research Digest community — including the short word descriptor, Easy Read Overviews, and text-to-speech audio. If you would like to help continue shaping the Digest, please don’t forget to complete the Research Digest feedback survey by clicking here.
Please note: The Research Digest shares current scientific findings for awareness and discussion. It is not a substitute for medical advice or treatment guidance, as much of the research featured is in its early stages and requires further confirmation.
You can join our community by signing up here to our mailing list and receiving the Digest straight to your inbox at the end of every month. We appreciate the support of everyone who reads the Digest – we encourage regular subscribers to support us with a monthly suggested donation of $2.
BIOLOGY
Temporal dynamics of the plasma proteomic landscape reveals maladaptation in ME/CFS following exertion
Authors: Germain A, Glass KA, Eckert MA, Giloteaux L, & Hanson MR (Cornell University, USA)
Publication: Molecular and Cellular Proteomics (December, 2025)
Link: https://www.sciencedirect.com/science/article/pii/S1535947625005663
Easy Read Overview: In this study, people with ME/CFS and healthy people completed two exercise tests on different days, and their blood was tested before and after each one. The results showed that people with ME/CFS had ongoing problems with their immune system, energy use, and muscle recovery after exercise, and these changes were linked to worse symptoms and lower exercise ability. The researchers concluded that PEM has a clear physical cause and that future ME/CFS research should look at changes over time and consider differences between males and females.
Post-exertional malaise (PEM) is frequently the most disabling symptom reported by ME/CFS patients. This study aimed to investigate the molecular underpinnings of PEM in people with ME/CFS, and to separate disease-specific molecular signatures of PEM from the physical deconditioning of sedentary controls.
132 participants were included in this study, which included 79 ME/CFS participants (Canadian Consensus Criteria) and 53 sedentary controls. All participants completed a 2-day Cardiopulmonary exercise test (CPET), with a 24-hour delay between each maximal effort exertion. Blood samples were taken before and after each CPET exertion (four samples in total).
Longitudinal plasma proteomic analysis of the blood samples found that ME/CFS patients showed persistent immune, metabolic, and neuromuscular dysregulation during the recovery phases of both tests. This included suppressed T and B cell signalling, and activation of pathways associated with glycolic stress. It was found that protein changes were associated with symptom severity, as well as reduced exercise capacity. Sex-specific variations were also noted.
The authors conclude that these findings support the physiological nature of PEM, including impaired molecular recovery from exertion. The authors propose that their findings demonstrate the value of dynamic, proteomic profiling over multiple time points as a method for identifying maladaptive exertion responses in ME/CFS. In addition, the authors demonstrate the need for sex-based biological variables when conducting ME/CFS research.
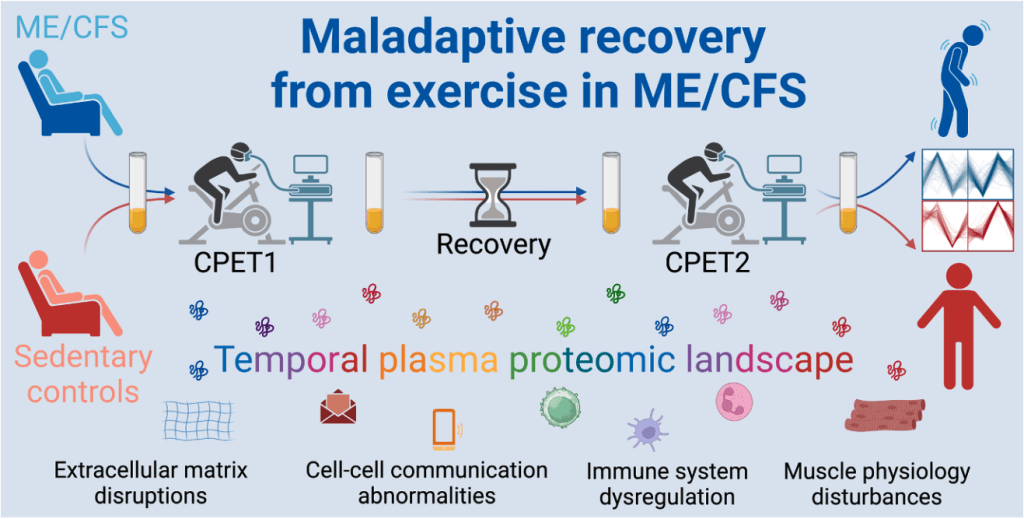
Figure: Graphical abstract. To study PEM, participants with ME/CFS and healthy controls underwent a two-day CPET. Plasma samples were taken before and after each exercise test, and proteomic analysis undertaken.
LIVED EXPERIENCE
Contested and neglected: Social and medical marginalization in severe Chronic Fatigue Syndrome
Author: Nezamdoust B, Ruel E, (Georgia State University, USA)
Publication: Social Science & Medicine (January, 2026)
Link: https://www.sciencedirect.com/science/article/pii/S0277953625010974
Easy Read Overview: ME/CFS has often been doubted and ignored by the medical system. About 25% of people with ME/CFS are severely ill, often bedbound and socially isolated, and they are hard to study because they cannot easily access care. This study looked at 342 tweets with the hashtag #severeME and found that people with severe ME/CFS face serious physical symptoms as well as emotional and social suffering, and are often shut out of healthcare and disability support. The authors say there is an urgent need for better medical recognition, stronger support systems, more research, and greater acceptance in society.
ME/CFS has a history of being medically-contested, stigmatised and neglected. As up to 80% of people with ME/CFS are women, this marginalisation is compounded by the medical field’s history of invalidating women’s experiences of symptoms, pain and illness severity.
Approximately 25% of people with ME/CFS have severe disease characterised by extreme loss of function, being bedbound, hypersensitivity, and social isolation. Combined with medical marginalisation, inaccessibility makes this population difficult to research, resulting in critical gaps in understanding the impact of the illness.
This study aimed to highlight the voices of people with severe ME/CFS through a qualitative analysis of 342 tweets using the hashtag #severeME, posted over two months in 2023. The researchers defined categories of tweet content related to the day-to-day lives of those with severe ME/CFS, which identified systemic barriers to critical support and wellbeing.
The study included 161 people (140 with severe ME/CFS, 12 caregivers, 9 unclear). Of these, 116 were women, 25 were men, 6 were non-binary, and gender was unknown for 14.
The researchers found that profound physical symptoms, debilitation and isolation were compounded by severe emotional, social and systemic suffering. These systemic issues resulted in exclusion from life-preserving care and disability systems, caused deep distress, and undermined quality-of-life.
Some people with severe ME/CFS avoided medical care to protect themselves from stigmatisation, dismissal and harmful advice. The authors found that families often became the final social safety net for those living with severe ME/CFS, leaving those without family support especially vulnerable. Severely impaired functioning and medical marginalisation leave this population unprotected by the systems meant to support those who are sick and disabled.
The authors conclude there is an urgent, critical need for medical validation and recognition, widespread structural reform, and research to underpin effective support, care and treatment, and to promote broader societal acceptance.
PROGNOSIS
Long COVID trajectories in the prospectively followed RECOVER-Adult US cohort
Authors: Thaweethai T, Donohue SE, Martin JN, Hornig M, Mosier JM, Shinnick DJ, … Levy BD(Harvard Medical School, USA)
Publication: Nature Communications (November 2025)
Link: https://doi.org/10.1038/s41467-025-65239-4
Easy Read Overview: Long COVID affects millions of people, but doctors are still learning how it changes over time. In this study from the National Institutes of Health’s RECOVER program, 3,659 adults were followed for 15 months and asked about their symptoms. The researchers found eight different symptom patterns, ranging from very severe and lasting symptoms to mild or no symptoms. They hope that understanding these patterns will help scientists find the causes of long COVID and develop better treatments.
While long COVID affects millions of people, its trajectory remains poorly defined. The adult cohort of the US National Institutes of Health’s Researching COVID to Enhance Recovery (RECOVER) initiative was used to define sub-groups and determine the long-term prognosis of long COVID.
For this study, 3659 participants were followed from the first infection through to a 15-month follow up. At the time of the study, Omicron was the dominant virus variant. Participants completed symptom surveys at 3, 6, 9, 12, and 15 months. Finite mixture models were used to identify longitudinal profiles. Participants with a Long COVID Research Index (LCRI) score of 11 or greater were classified as having long COVID.
The authors identified eight distinct profiles.
Profile A: persistent, high symptom burden. Most participants whose LCRI was very high at three months belonged to this profile. Profile B: intermittently high symptom burden. Profile C: improving, moderate symptom burden. Profile D: improving, low symptom burden. Profile E: worsening, moderate symptom burden. Profile F: delayed worsening symptom burden. This profile was driven by an increase in post-exertional malaise (PEM) after month 12. Profile G: consistent, low symptom burden. Profile H: consistent, minimal to no symptom burden., This was the largest proportion of the cohort.
Of the 3659 participants, only 377 met the long COVID threshold at three months post-infection: 46% were classified as profile A, 35% as profile B, 18% as profile C, and 1% as profile D. Those in the “worsening’ profiles (E and F), had only slightly higher rates of COVID-19 reinfection than other profiles. This suggests that it is unlikely that increased their LCRI is due to by newly developed long COVID.
The authors highlight the strength of the RECOVER study in its use of an unbiased population-based cohort and frequent, serial measurements. They suggest this enabled the identification of a greater number of distinct profiles compared to previous studies. However, they acknowledge that participants with the highest LCRIs may have been less likely to return for follow-up due to high symptom burden, potentially resulting in missing data. They also note that long COVID in the Omicron era may present with different profiles compared to other variants. Nonetheless, they argue that identifying these distinct profiles should facilitate the discovery of biomarkers and risk factors.
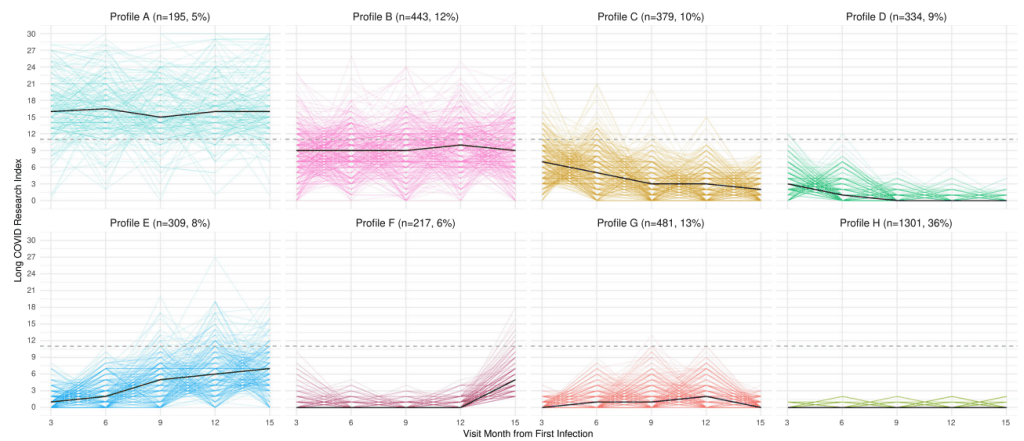
Figure: Finite mixture models were used to identify distinct trajectories of the Long COVID Research Index between 3 and 15 months after the first SARS-CoV-2 infection. The median (solid black line) and individual trajectories (solid colored lines) are shown for the 8 longitudinal profiles (A–H) identified. The Long COVID Research Index threshold of 11 is provided (dashed line).
BIOLOGY
Chronic fatigue syndrome new research
Authors: Young, B.
Publication: ABC Nightlife
Link: https://www.abc.net.au/listen/programs/nightlife/cfs-research/106268112
Professor Sonya Marshall-Gradisnik and Professor Peter Smith were interviewed by Bern Young on ABC Nightlife about the publication of recent research by the National Centre for Neuroimmunology and Emerging Diseases (NCNED). This study involved a replication of NCNED’s research by an independent research group in Western Australia, confirming a biological mechanism in ME/CFS.
The segment runs for 26 minutes.

